 
Radium (Latin radius, ray) was discovered by Maria Skłodowska-Curie and her husband Pierre in 1898 in pitchblende/ uraninite from North Bohemia (area around Jáchymov). Spas featuring radium-rich water are still occasionally touted as beneficial, such as those in Misasa, Tottori, Japan.Such products soon fell out of vogue, and were prohibited by authorities in many countries, after it was discovered they could have real and serious adverse health effects. At the turn of the 20th century radium was a popular additive in products like toothpaste, hair creams, and even food items due to its supposed curative powers.One unit for radioactivity, the non- SI curie, is based on the radioactivity of radium-226 (see Radioactivity).Radium-223 is currently under investigation for use in medicine as cancer treatment of bone metastasis.Radium was also put in many foods for taste and a preservative, but also exposed many people to radiation.Radium (usually in the form of radium chloride) is used in medicine to produce radon gas which in turn is used as a cancer treatment.When mixed with beryllium it is a neutron source for physics experiments.Although tritium's beta radiation is potentially dangerous if ingested, it has replaced radium in these applications. Objects painted with this paint may still be dangerous, and must be handled properly. Radium was still used in dials as late as the 1950's. Soon afterward, the adverse effects of radioactivity became widely known. More than 100 former watch dial painters who used their lips to shape the paintbrush died from the radiation. Formerly used in self-luminous paints for watches, aircraft switches, clocks, and instrument dials.More recently discovered radioisotopes, such as cobalt-60 and caesium-137, are replacing radium in even these limited uses because several of these are much more powerful and others are safer to handle. 
Some of the practical uses of radium are derived from its radiative properties. Radium is luminescent (giving a faint blue colour), reacts violently with water to form radium hydroxide and is slightly more volatile than barium. When freshly prepared, pure radium metal is brilliant white, but blackens when exposed to air (probably due to nitride formation). Radium also produces neutrons when mixed with beryllium. Radium preparations are remarkable for maintaining themselves at a higher temperature than their surroundings, and for their radiations, which are of three kinds: alpha particles, beta particles, and gamma rays. This metal is found in minute quantities in the uranium ore pitchblende, and various other uranium minerals. The heaviest of the alkaline earth metals, radium is intensely radioactive and resembles barium in its chemical behaviour. 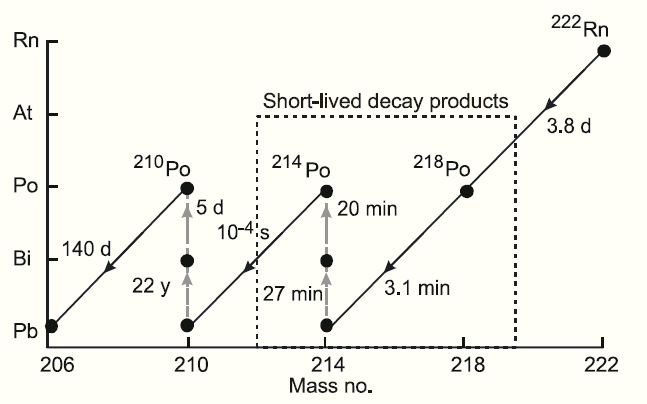
Its most stable isotope, 226Ra, has a half-life of 1602 years and decays into radon gas. Radium is an alkaline earth metal that is found in trace amounts in uranium ores. Its appearance is almost pure white, but it readily oxidizes on exposure to air, turning black. Radium ( IPA: /ˈreɪdiəm/) is a chemical element, which has the symbol Ra and atomic number 88 (see the periodic table). 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
